 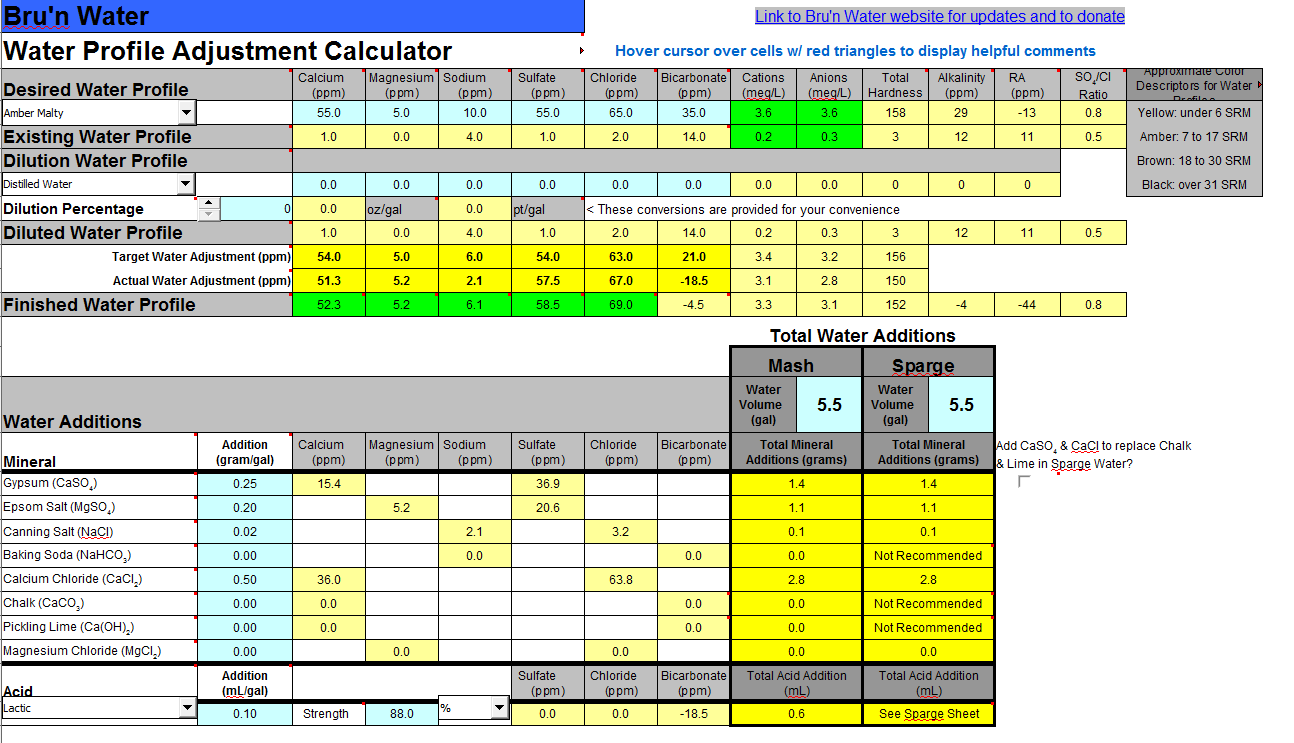
Table Salt – Table salt contributes sodium and chloride.1 teaspoon in five gallons will increase sulfate by 100 ppm and magnesium by 25 ppm. Magnesium Sulfate – As the name suggest, magnesium sulfate adds both magnesium and sulfate.For every teaspoon added to 5 gallons of water, it adds about 60 ppm of carbonate and 36 ppm of calcium. Calcium Carbonate – Calcium carbonate raises pH.1 teaspoon per five gallons adds 140 ppm of sulfate and 60 ppm of calcium. Gypsum – Gypsum adds calcium sulfate, which lowers pH.Once the concentration of the minerals listed above are established, a brewer can use the following to make adjustments: There may be a balancing act between maintaining an appropriate pH level and replicating the water of a particular region. Once you’ve established a baseline, you can add minerals to adjust your water profile. This would be the purist way of handling your brewing water chemistry. Another option is to use only reverse osmosis or distilled water, in which case you’re essentially starting from pure H 2O. You can also check with the local water department for a water quality report. Someone may have already figured out the mineral content of your local water source and offer some recommendations for how to adjust your water, if at all. Consider checking with your local homebrew club. 
To recreate a Munich Dunkel or English Bitter, it may be necessary to replicate the brewing water chemistry of those locations.īrewing water chemistry analysis and profiles for specific locations is widely available from books such as Homebrewing for Dummies, Designing Great Beers, and others.īefore making changes to your brewing water, it’s very important to establish a starting point, otherwise you’re just shooting in the dark. Some styles of beer were developed based on the local water profiles of the breweries that made them. Water Chemistry for Brewing Certain Styles Depending on the source water, it may be necessary to add mineral salts to raise or lower the brewing water’s pH for a given beer recipe. Certain malted barleys can lower pH more than others. Pure water has a pH of about 7, whereas ideal conditions for enzyme activity is about 5.4-5.5. The enzymes work best when working within a specific pH range. One of the primary factors is pH, or acidity. 
The enzymes in malt need certain conditions in order to covert starches into sugars. This is particularly important for all-grain brewers, but also partial mash brewers. One of the many ways water chemistry comes into play in home brewing is with the mashing process. Sulfate – Sulfate can introduce harsh, dry flavors.Carbonate/Bicarbonate – These can slow the mashing process and make hop flavors taste harsh.Sodium – Sodium can help give beer body, but too much might make a beer taste salty.Magnesium – Lowers pH and aids yeast health.Calcium – Lowers pH and helps with mash conversion.When looking at the brewing water chemistry, brewers pay attention to the following minerals: For these reasons, many home brewers pay fierce attention to the chemistry profile of the water they use for brewing. But in addition to that, the concentration of minerals in your brewing water can impact not only flavor, but also mash performance, acidity, hop bitterness, yeast health, body, mouthfeel, and other factors. For one, it has to be clean and free of microbiological contamination. In home brewing, water chemistry can have a huge impact on beer flavor and quality. 
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
